Abstract
Purpose
Drug-coated balloons (DCBs) for femoropopliteal peripheral artery disease have been shown to be clinically superior and cost-effective compared to conventional percutaneous transluminal angioplasty (PTA). However, few studies enrolled patients with chronic limb-threatening ischemia (CLTI). Our objective was to study the cost-effectiveness of endovascular treatment with versus without DCB in CLTI patient populations in the Netherlands and Germany.
Material and Methods
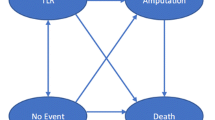
Target lesion revascularization (TLR) and major amputation rates were obtained from the CLTI subgroup of the IN.PACT Global study. Rates for “status quo” treatment involving PTA with primary or bailout stenting were derived from systematic literature search. Costs and cost-effectiveness were calculated using a decision-analytic Markov model considering, in the base case, a 2-year horizon, and strategy-specific quality-adjusted life year (QALY) gains calculated from survival and health state-specific utilities. A willingness-to-pay threshold of €50,000/QALY was assumed, and extensive sensitivity analyses were performed.
Results
Model-projected 24-month probabilities of TLR were 26.2% and 32.8% for treatment with and without DCB, and probabilities for amputation were 2.8% and 11.9%, respectively. DCB added 0.017 QALYs while saving €1,030 in the Dutch setting and €513 in the German setting, respectively. DCB was found dominant or cost-effective across a wide range of assumptions.
Conclusion
Urea excipient drug-coated balloon therapy for treating CLTI from femoropopliteal artery disease is associated with improved patient outcomes and expected overall cost savings to payers in the Dutch and German healthcare systems, rendering it a cost-effective and likely dominant treatment strategy.


Similar content being viewed by others
References
Attema AE, Brouwer WBF, Claxton K. Discounting in economic evaluations. Pharmacoeconomics. 2018;36(7):745–58.
Bausback Y, Willfort-Ehringer A, Sievert H, et al. Six-month results from the initial randomized study of the ranger paclitaxel-coated balloon in the femoropopliteal segment. J Endovasc Ther. 2017;24(4):459–67.
Biagioni RB, Nasser F, Matielo MF, et al. Comparison of bypass and endovascular intervention for popliteal occlusion with the involvement of trifurcation for critical limb ischemia. Ann Vasc Surg. 2020;63:218–26.
Bosma J, Vahl A, Wisselink W. Systematic review on health-related quality of life after revascularization and primary amputation in patients with critical limb ischemia. Ann Vasc Surg. 2013;27(8):1105–14.
Bruhn D, Martin AA, Tavares R, Hunt B, Pollock RF. Cost-utility of albiglutide versus insulin lispro, insulin glargine, and sitagliptin for the treatment of type 2 diabetes in the US. J Med Econ. 2016;19(7):672–83.
Dua A, Lee CJ. Epidemiology of Peripheral Arterial Disease and critical limb ischemia. Tech Vasc Interv Radiol. 2016;19(2):91–5.
Farber A, Rosenfield K, Siami FS, Strong M, Menard M. The BEST-CLI trial is nearing the finish line and promises to be worth the wait. J Vasc Surg. 2019;69(2):470–81.
Goksel OS, Karpuzoğlu E, Işsever H, Çinar B. Midterm results with drug-coated balloons for SFA lesions in patients with CLI: comparison with conventional bypass surgery. Int Angiol. 2018;37(5):365–9.
InEK (2020) Fallpauschalen-Katalog 2020 (German-Diagnosis Related Groups-System). Institut für das Entgeltsystem im Krankenhaus (InEK GmbH), https://www.g-drg.de/aG-DRG-System_2020/Fallpauschalen-Katalog/Fallpauschalen-Katalog_2020.
Jia X, Zhang J, Zhuang B, et al. Acotec drug-coated balloon catheter: randomized, multicenter, controlled clinical study in femoropopliteal arteries: evidence from the AcoArt I trial. JACC Cardiovasc Interv. 2016;9(18):1941–9.
Katsanos K, Geisler BP, Garner AM, Zayed H, Cleveland T, Pietzsch JB. Economic analysis of endovascular drug-eluting treatments for femoropopliteal artery disease in the UK. BMJ Open. 2016;6(5):e011245.
Leon MB, Piazza N, Nikolsky E, et al. Standardized endpoint definitions for transcatheter aortic valve implantation clinical trials: a consensus report from the valve academic research consortium. Eur Heart J. 2011;32(2):205–17.
Marseille E, Larson B, Kazi DS, Kahn JG, Rosen S. Thresholds for the cost-effectiveness of interventions: alternative approaches. Bull World Health Organ. 2015;93(2):118–24.
Meng FC, Chen PL, Lee CY, Shih CC, Chen IM. Real-world comparison of drug-eluting and bare-metal stents in superficial femoral artery occlusive disease with trans-atlantic intersociety consensus B lesions: a 2-year. Single-Inst Study Acta Cardiol Sin. 2018;34(2):130–6.
Phair J, Carnevale M, Lipsitz EC, Shariff S, Scher L, Garg K. Amputation-free survival in patients with critical limb ischemia treated with paclitaxel-eluting stents and paclitaxel-coated balloons. Ann Vasc Surg. 2020;62:8–14.
Pietzsch JB, Geisler BP, Garner AM, Zeller T, Jaff MR. Economic analysis of endovascular interventions for femoropopliteal arterial disease: a systematic review and budget impact model for the United States and Germany. Catheter Cardiovasc Interv. 2014;84(4):546–54.
Popplewell MA, Davies HOB, Narayanswami J, et al. A comparison of outcomes in patients with infrapopliteal disease randomised to vein bypass or plain balloon angioplasty in the bypass versus angioplasty in severe ischaemia of the leg (BASIL) trial. Eur J Vasc Endovasc Surg. 2017;54(2):195–201.
Reijnen M, van Wijck I, Zeller T, et al. Outcomes after drug-coated balloon treatment of femoropopliteal lesions in patients with critical limb ischemia: a post hoc analysis from the IN.PACT global study. J Endovasc Ther. 2019;26(3):305–15.
Salisbury AC, Li H, Vilain KR, et al. Cost-effectiveness of endovascular femoropopliteal intervention using drug-coated balloons versus standard percutaneous transluminal angioplasty: results from the IN.PACT SFA II trial. JACC Cardiovasc Interv. 2016;9(22):2343–52.
Santos Á, Valdivia A, Alguacil S, Sanus E, Guaita J, Zúñiga C. One year results in the endovascular treatment of the femoro-popliteal segment with the Tigris® stent in patients with critical limb ischemia. Eur J Vasc Endovasc Surg. 2019;58:e466.
Scheinert D, Schmidt A, Zeller T, et al. German center subanalysis of the LEVANT 2 global randomized study of the lutonix drug-coated balloon in the treatment of femoropopliteal occlusive disease. J Endovasc Ther. 2016;23(3):409–16.
Tepe G, Laird J, Schneider P, et al. Drug-coated balloon versus standard percutaneous transluminal angioplasty for the treatment of superficial femoral and popliteal peripheral artery disease: 12-month results from the INPACT SFA randomized trial. Circulation. 2015;131(5):495–502.
Todoran TM, Connors G, Engelson BA, Sobieszczyk PS, Eisenhauer AC, Kinlay S. Femoral artery percutaneous revascularization for patients with critical limb ischemia: outcomes compared to patients with claudication over 2.5 years. Vasc Med. 2012;17(3):138–44.
Torres-Blanco A, Edo-Fleta G, Gomez-Palones F, Molina-Nacher V, Ortiz-Monzon E. Mid-term outcomes of endovascular treatment for TASC-II D femoropopliteal occlusive disease with critical limb ischemia. Cardiovasc Intervent Radiol. 2016;39(3):344–52.
Nederlandse Zorgautoriteit. dbc-informatiesysteem (DIS); https://www.opendisdata.nl/dis/over, 2020.
Funding
This research was—in part—funded by Medtronic, Inc. The authors retained the right to publish without approval of the funding source.
Author information
Authors and Affiliations
Contributions
JBP, BPG, and MMPJR contributed to conception, design, analysis, and interpretation and wrote the article. SH, IPSW, MMPJR, ARI, BPG, and JBP collected the data. JBP and BPG helped in model development. JBP, BPG, ARI, SH, IPSW, and MMPJR critically revised the article and provided final approval of the article. JBP and MMPJR had overall responsibility.
Corresponding author
Ethics declarations
Conflict of interest
Jan B. Pietzsch, Ph.D.: Dr. Pietzsch is the president, CEO, and a shareholder of Wing Tech Inc., an independent research firm conducting health economic analyses. Wing Tech Inc. received consulting fees from Medtronic, Inc. to conduct the analyses underlying this study. Benjamin P. Geisler, M.D., M.P.H.: Dr. Geisler is a senior consultant for Wing Tech Inc., an independent research firm conducting health economic analyses. Wing Tech Inc. received consulting fees from Medtronic, Inc. to conduct the analyses underlying this study. Annabelle R. Iken, M.Sc.: Mrs. Iken is a research associate for Wing Tech Inc., an independent research firm conducting health economic analyses. Wing Tech Inc. received consulting fees from Medtronic, Inc. to conduct the analyses underlying this study. Iris van Wijck, BSc: No conflict of interest. Suzanne Holewijn, MSc, PhD: No conflict of interest. Michel M. P. J. Reijnen, MD, PhD: Dr. Reijnen received consulting fees and research funding from Medtronic, Inc.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pietzsch, J.B., Geisler, B.P., Iken, A.R. et al. Cost-Effectiveness of Urea Excipient-Based Drug-Coated Balloons for Chronic Limb-Threatening Ischemia from Femoropopliteal Disease in the Netherlands and Germany. Cardiovasc Intervent Radiol 45, 298–305 (2022). https://doi.org/10.1007/s00270-021-03050-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-021-03050-6